The pros and cons of rapid testing for COVID-19
Many have been complaining that the standard PCR test to detect the new coronavirus is too slow when it comes to results. In a lot of critical cases, the results come too late, meaning the patient had already died of COVID-19 complications.
There is a quicker alternative to the standard PCR test: it’s called “rapid antibody testing” and a lot of people, including government agencies are using them to help spot more potential cases. However, we must take note that these rapid test kits do not actually detect if a person has the COVID-19 virus or not. Only the PCR test can detect the virus.
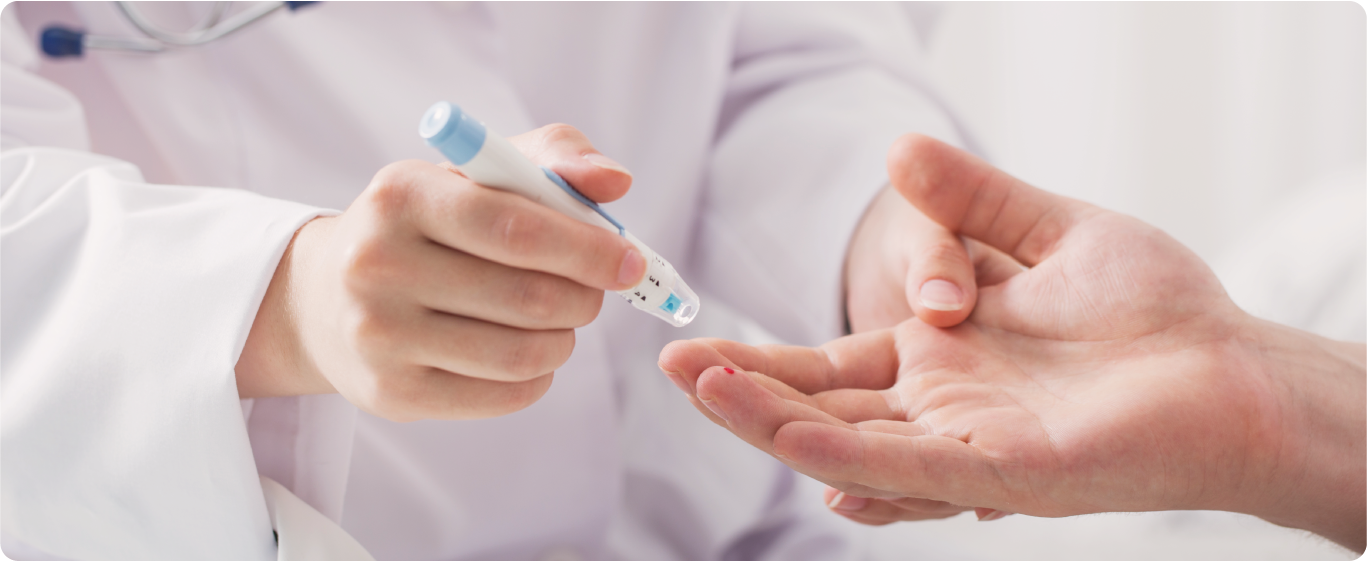
The rapid antibody test [1] requires a blood sample. Instead of detecting the presence of the actual virus, the rapid test can only detect the presence of antibodies [2] specific to the virus.
What does the presence of these antibodies mean? [3] It’s a sign that the person a) is currently infected; or b) the person was previously infected [4] and has now recovered. The processing time for this test will only take approximately 15 to 45 minutes.
Unlike the rapid antibody tests, PCR tests use actual swabs from patients taken from the nose or throat. This test takes longer before results are released; from 24 hours or up to 3 days for the results to be released. [1]
It’s been proposed that this rapid testing be done on workers and employees returning to work once the lockdowns are relaxed. Among the advantages of rapid testing is cost, as it is much cheaper to use, and the speed of results. But there’s a trade-off involved.
Testing limitations
Many medical experts don’t recommend the antibody testing kits because of their “high level of inaccuracies, particularly the high prevalence of “false positives.” [5] The World Health Organization does not recommend using rapid diagnostic tests to confirm COVID-19 cases. [6]
Despite these worries, the government decided to use rapid test kits in conjunction with PCR-based test kits. While rapid tests aren’t reliable enough for diagnostic purposes, they can be useful in screening those who are possibly vulnerable to the disease. [7]
The Project Antibody Rapid Test Kit, a private sector-led initiative, also suggests that combining both rapid and PCR-based tests will help the economy to restart and provide a safety net against a second wave of COVID-19 cases. [8]
For now, only the following persons can be tested using the rapid testing kits: [9]
-
Asymptomatic healthcare workers but have “unprotected exposure”
-
A person who shows symptoms and had close contact of a known or suspected case
-
Asymptomatic person who had close contact of a COVID-19 patient
-
Filipinos returning from another country whether symptomatic or asymptomatic
-
Asymptomatic people but are residing in areas with suspected community-based COVID-19 transmission
Now, as we slowly adapt to the “new normal,” we can only rely on our available resources and continue to follow preventive measures.
---
References:
-
Magsambol B. Fast Facts: What’s the difference between PCR, rapid antibody tests?; Rappler. [Internet] 2020 [cited 28 May 2020]. Available from: https://www.rappler.com/newsbreak/iq/261869-things-to-know-polymerase-chain-reaction-rapid-anti-body-tests.
-
Centers for Disease Control and Prevention. Serology Testing for COVID-19 [Internet] 2020 [cited 28 May 2020]. Available from https://www.cdc.gov/coronavirus/2019-ncov/lab/serology-testing.html.
-
Sabillo K. Doctors group in PH warn vs. COVID-19 rapid tests to screen returning workers. ABS-CBN News. [Internet] 2020 [cited 28 May 2020]. Available from: https://news.abs-cbn.com/news/05/19/20/doctors-groups-in-ph-warn-vs-covid-19-rapid-tests-to-screen-returning-workers.
-
Labzin L. Why can’t we use antibody tests for diagnosing COVID-19 yet?; The Conversation. [Internet] 2020 [cited 28 May 2020]. Available from: https://theconversation.com/why-cant-we-use-antibody-tests-for-diagnosing-covid-19-yet-138519
-
Crisostomo S. Medical Groups Reiterate Opposition to Rapid Test Kits for Workers. One News PH. [Internet] 2020 [cited 28 May 2020]. Available from: https://www.onenews.ph/medical-groups-reiterate-opposition-to-rapid-test-kits-for-workers
-
Cabico G. RITM to assess reliability of FDA-approved rapid test kits. PhilStar Global. [Internet] 2020; [cited 28 May 2020] Available from: https://www.philstar.com/headlines/2020/04/21/2008801/ritm-assess-reliability-fda-approved-rapid-test-kits.
-
Tomacruz S. EXPLAINER: What to expect from the Philippines' expanded coronavirus testing. Rappler [Internet] 2020 [cited 28 May 2020]. Available from: https://www.rappler.com/newsbreak/iq/258723-explainer-philippines-expanded-coronavirus-testing
-
Desiderio L. Rapid testing with PCR seen to help restart economy safely. PhilStar Global. [Internet] 2020 [cited 28 May 2020]. Available from:https://www.philstar.com/headlines/2020/05/24/2016113/rapid-testing-pcr-seen-help-restart-economy-safely.
-
Tan L. List: Who can be tested using rapid COVID-19 test kits?. CNN Philippines. [Internet] 2020 [cited 28 May 2020]; Available from: https://www.cnnphilippines.com/news/2020/4/20/Rapid-antibody-test-kits-COVID-19-Philippines.html.

